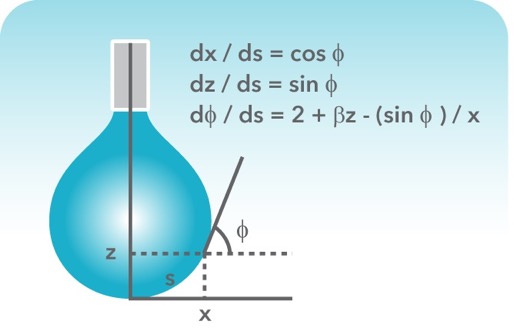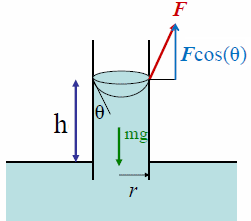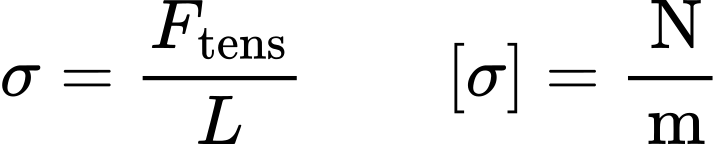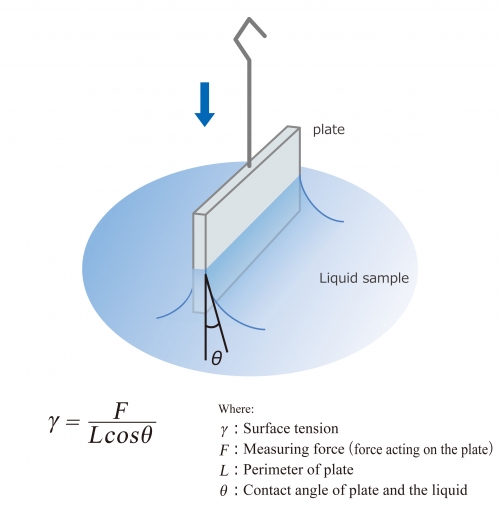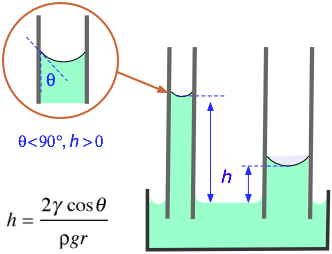
How would you calculate the surface tension of a 2% (w/v) solution of a wetting agent that has a density of 1.008 g/mL and that rises 6.6 cm in a capillary tube
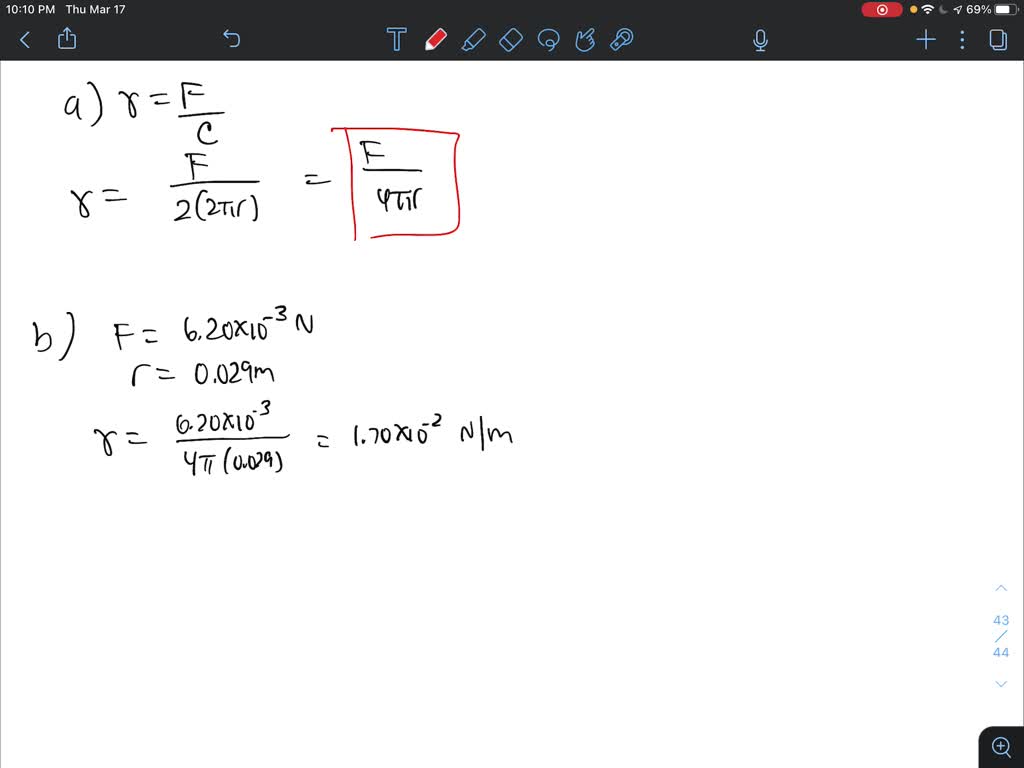
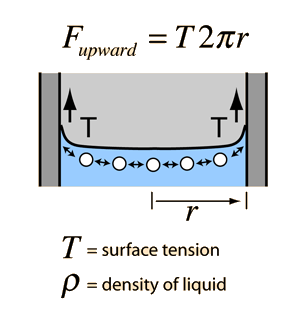
SOLVED: The surface tension of a liquid can be determined by measuring the force F needed to just lift a circular platinum ring of radius r from the surface of the liquid.